ISRA FACTSHEETS
ISRA FACTSHEETS
AUSTRALIA AND SOUTHEAST INDIAN OCEAN
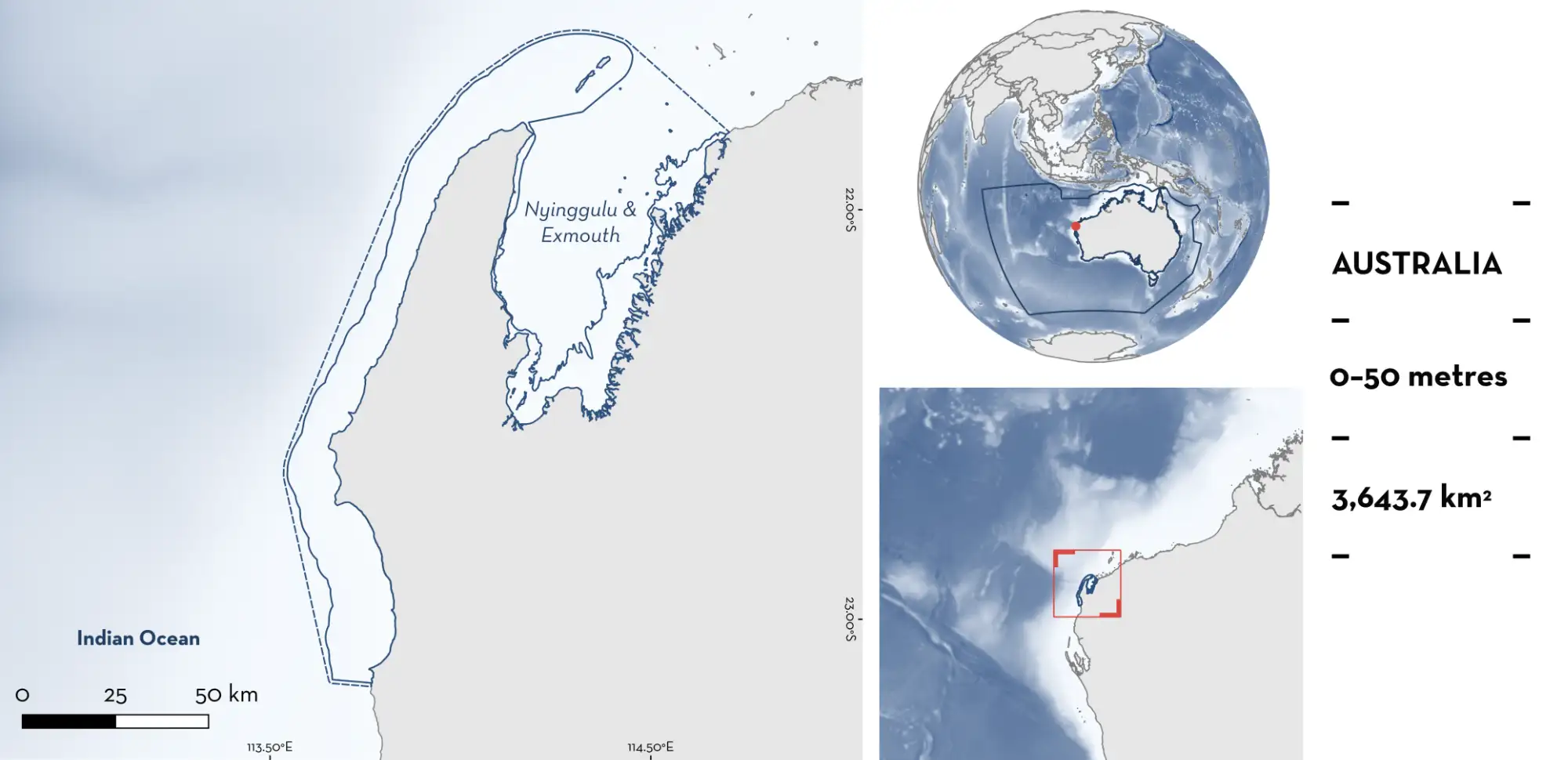
Nyinggulu & Exmouth
Summary
Nyinggulu & Exmouth is located in the Gascoyne region of Western Australia, Australia. It is situated in Nyinggulu Ganyarjarrri, Thalanyji, Baiyungu, and Yinigurdira Countries. This area encompasses the Ningaloo Lagoon, the outer reef, and Exmouth Gulf. The habitat is characterised by shallow sand flats, coral reefs, cyanobacterial mats, seagrass beds, mangroves, mudflats, macroalgae, and pelagic habitats. This area overlaps with a Key Biodiversity Area and two marine protected areas. Within this area there are: threatened species (e.g., Sharptooth Lemon Shark Negaprion acutidens); reproductive areas (e.g., Australian Whipray Himantura australis); feeding areas (e.g., Giant Guitarfish Glaucostegus typus); undefined aggregations (e.g., Bluespotted Lagoon Ray Taeniura lymma); distinctive attributes (Grey Reef Shark Carcharhinus amblyrhynchos); and the area sustains a high diversity of sharks (39 species).
Download factsheet
Nyinggulu & Exmouth
DESCRIPTION OF HABITAT
Nyinggulu & Exmouth is located in the Gascoyne region of Western Australia, Australia. It is situated in Nyinggulu Ganyarjarrri, Thalanyji, Baiyungu, and Yinigurdira Countries. This area spans from Bateman Bay (Coral Bay) to Urala Creek (south of Onslow), including Skeleton Bay, Cardabia Passage, Asho’s Gap, Point Cloates, Winderabandi Point, Mangrove Bay, Pelican Point, Bay of Rest, and Giralia Bay. It encompasses the Ningaloo Lagoon, outer reef, and Exmouth Gulf. The Ningaloo Lagoon is a shallow coastal system bordered offshore by one of the world’s longest and most extensive fringing reefs (Vanderklift et al. 2020). The lagoon supports a mosaic of sandy benthos, rocky reefs, seagrass and macroalgal meadows, coral outcrops, and a small mangrove system (Vanderklift et al. 2020). The outer reef is situated along the reef edge outside the lagoon with a substrate composed of a limestone platform that transitions to sediment-covered shelf beyond 35 m (Vanderklift et al. 2020). Exmouth Gulf is the only sheltered embayment in the Pilbara, and one of the few in Western Australia. It is a wide, shallow, inverse estuary with a wide intertidal flat with habitats characterised by seagrass beds, mangroves, mudflats, macroalgae, and cyanobacterial mats (WAMSI 2025).
Sea surface temperatures (SST) vary from ∼24–26°C on average in Ningaloo (Falter et al. 2014). In Exmouth Gulf, SST varies between ~30°C (austral summer) and ~19–20°C (winter) (WAMSI 2025). The region is hot and arid with freshwater runoff from land being low. Rainfall is highly variable and largely tied to the summer monsoon season (November–April), and tropical cyclones, which can bring heavy downpours, flooding, and freshwater pulses, especially into the gulf (Vanderklift et al. 2020). The outer reef is influenced by the Leeuwin Current that drives higher rates of pelagic production here compared to most other parts of Western Australia (Hanson et al. 2005; Rousseaux et al. 2012). The system is characterised by elevated productivity during autumn (March–May) and winter (June–August), when upwelling along the reef front near the continental shelf interacts with ocean currents (Hanson et al. 2005; Sleeman et al. 2010; Vanderklift et al. 2020). Productivity peaks as the southward-flowing, warm Leeuwin Current intensifies alongside the cooler, northward-flowing Ningaloo Current (Wilson et al. 2001; Hanson et al. 2005). These dynamics generate strong oceanographic fronts near reef passages, where dense lagoon waters mix with fresher shelf waters, supporting plankton assemblages (Wilson et al. 2001, 2002; Hanson et al. 2005). Offshore, mesoscale eddy activity further enhances productivity by driving localised upwelling that sustains high primary production (Woo et al. 2006; Xu et al. 2013; D’Antonio et al. 2024).
This area overlaps with the Exmouth Gulf Mangroves Key Biodiversity Area (KBA 2025), Ningaloo Marine Park, and Exmouth Gulf Marine Park (WA DBCA 2025).
This Important Shark and Ray Area is benthic and pelagic and is delineated from inshore and surface waters (0 m) to 50 m based on the depth range of Qualifying Species in the area.
CRITERION A
VULNERABILITY
Thirty-four Qualifying Species considered threatened with extinction according to the IUCN Red List of Threatened Species regularly occur in the area. Threatened sharks comprise three Critically Endangered species, five Endangered species, and ten Vulnerable species; threatened rays comprise five Critically Endangered species, seven Endangered species, and four Vulnerable species (IUCN 2025).
CRITERION C
SUB-CRITERION C1 – REPRODUCTIVE AREAS
Nyinggulu & Exmouth is an important reproductive area for three shark and four ray species.
Ningaloo & Exmouth Coast ISRA within the Nyinggulu & Exmouth area is important for the reproduction of Nervous Shark, Blacktip Reef Shark, Sharptooth Lemon Shark, Giant Guitarfish, Australian Whipray, Broad Cowtail Ray, and Green Sawfish. Several studies, combined with unpublished data and citizen science, report large numbers of neonates, young-of-the-year (YOY), and older juveniles of these species regularly and predictably occurring in this area. Fishery-independent gillnet surveys (150 mm mesh size, 60 m length) and visual observations from vessel-based surveys were conducted in southwestern Exmouth Gulf in 2021–2022 (RL Bateman-John unpubl. data 2022), in southwestern and eastern Exmouth Gulf in 2024–2025 (Morgan et al. 2020; KO Lear et al. unpubl. data 2025), in Urala Creek North and South during February 2019 (Morgan et al. 2020), and in the southeastern Gulf at Giralia in August 2020 (DL Morgan et al. unpubl. data 2020). In 2024 and 2025, aerial drone surveys (flown at 2 m/s and 20 m height) were also conducted across the southwestern and eastern Exmouth Gulf (S Gudge & K Kliska unpubl. data 2025). All of these sites are located in this area.
In January 2022, in the Bay of Rest in this area, ten adult female Nervous Sharks (>91 cm total length; TL) were captured. Four of these (104–117 cm TL; 40% of adult females) bore bite marks around the pectoral fins or body, most likely mating scars (RL Bateman-John unpubl. data 2025). This suggests that this area is used for reproductive behaviour. In addition, Nervous Sharks are the most common shark species encountered in shallow areas of Exmouth Gulf: between 2024–2025, they comprised 84% of shark catches during gillnet surveys (150 mm mesh) in the southern and eastern Exmouth Gulf (KO Lear et al. unpubl. data 2025). Although YOY (<60 cm TL) are not readily caught in 100–150 mm mesh size gillnets, 18 were captured during these surveys (KO Lear et al. unpubl. data 2025). Further, one neonate or YOY (44 cm TL) was captured in the Bay of Rest in 2022 (RL Bateman-John unpubl. data 2025). Neonates and YOY are also often observed during vessel-based surveys, including at least five individuals estimated at 30–60 cm TL in the Bay of Rest from 2021–2022, and 65 individuals likely to be a mixture of YOY and older juveniles across the southern and eastern Exmouth Gulf in 2024–2025 (KO Lear et al. unpubl. data 2025). Aggregations of neonate Nervous Sharks (~40–50 cm TL) were observed in Urala Creek North and South in 2019 (Morgan et al. 2020). Nervous Shark size-at-birth is 35–40 cm TL (Ebert et al. 2021), and YOY are estimated as those <60 cm TL (White et al. 2002). Size-at-maturity is estimated at 91–105 cm TL for females and 89–95 cm TL for males (White et al. 2002). Combined, this evidence indicates that this area is important for various life-stages of this species.
Between November 2012 and January 2014, Blacktip Reef Sharks were captured in shallow water with gillnets and rod-and-line in Mangrove Bay in this area (Oh et al. 2017). Blacktip Reef Sharks (n = 13) ranged between 51–107 cm TL (mean = 63.9 cm TL). Most of the captured sharks were neonates or YOY with umbilical scars in various stages of healing (age <1 year), except for one Blacktip Reef Shark that was a juvenile female (Oh et al. 2017). Furthermore, in 2021–2022 and 2024–2025, gillnet surveys (100–150 mm mesh size, 60 m length) captured Blacktip Reef Sharks of various body sizes, although YOY are not readily caught with the mesh size used in these surveys. Six YOY (<70 cm TL) were visually observed during gillnet surveys in the Bay of Rest in 2021–2022 (RL Bateman-John unpubl. data 2022). In 2024–2025, seven Blacktip Reef Sharks were captured in gillnets in the northeastern and southwestern Exmouth Gulf, including three YOY (<70 cm TL) (KO Lear et al. unpubl. data 2025). An additional 18 Blacktip Reef Sharks were visually observed during these surveys; 16 were small and likely YOY or older juveniles (KO Lear et al. unpubl. data 2025). In Bundegi, aggregations of over 20 YOY Blacktip Reef Sharks are commonly observed. These groups have appeared during every recreational drone survey undertaken between 2018 and 2025, with at least 10 confirmed observation events (KO Murphy unpubl. data 2025). In northeastern Australia, size-at-birth, YOY, and age-1 individuals for Blacktip Reef Shark range from 58–67 cm TL, 58–73 cm TL, and 62–85 cm TL, respectively (Chin et al. 2013). Here, YOY are estimated as those <70 cm TL.
In March, November, and December 2012, Sharptooth Lemon Sharks (n = 23) were captured in shallow water with gillnets and rod-and-line and fitted with internal acoustic transmitters in Mangrove Bay in this area (Oh et al. 2017). Animals measured between 63–101 cm TL (mean = 75.2 cm TL) and were neonates or YOY with umbilical scars in various stages of healing (age <1 year). Umbilical scars and captures of neonate (<1 week old) Sharptooth Lemon Sharks between November–March indicate local pupping and extended occupancy (up to 17 months). Size-at-birth for Sharptooth Lemon Shark is 45–80 cm TL (Ebert et al. 2021). Tagged sharks were monitored for 2–544 days between March 2013 and May 2015 by 85 receivers across the northern Ningaloo Reef Marine Park: 71 receivers in Mangrove Bay and two cross-shelf lines of eight receivers at Tantabiddi and seven receivers at Turquoise Bay. Shark showed high long-term residency (>30 days) with small activity spaces (mean 95% kernel area = 4.5 km2) (Oh et al. 2017). In addition, between 2012–2018, 38 adult Sharptooth Lemon Sharks (23 females, 240–280 cm TL; 18 males, 234–266 cm TL) were captured using single circle-hooks and tagged in Mangrove Bay (Pillans et al. 2021). The acoustic receiver network spanned ~150 km of coastline and comprised five sites, with most located within this area, except Coral Bay. Adults exhibited long-term site fidelity, with 79% of detections within Mangrove Bay. Pregnant, post-partum, and recently mated females were detected in successive years, confirming Mangrove Bay as a key reproductive habitat (Pillans et al. 2021). In 2021–2022 and 2024–2025, gillnet surveys (150 mm mesh size, 60 m length) captured 80 Sharptooth Lemon Sharks, with an additional 53 individuals observed during vessel-based surveys in shallow waters (RL Bateman-John unpubl. data 2022; KO Lear et al. unpubl. data 2025). Of the 80 captured, 42 (52%) were <80 cm TL, and 39 (73%) visually observed Sharptooth Lemon Sharks were estimated to be <80 cm TL, including an aggregation of 13 individuals schooling in the shallows. YOY were caught in each year surveyed. In addition, in 2024–2025, a total of 384 Sharptooth Lemon Sharks were identified from drone surveys across shallow areas in the eastern and southern Exmouth Gulf (K Kliska & S Gudge unpubl. data 2025). Size of these sharks was not estimated during drone analyses, but given the shallow habitat surveyed and the dominance of YOY among captured sharks during the gillnet surveys, it is likely that the majority were YOY. Groups of up to five individuals were observed during the aerial surveys. As no growth data are available for this species, the growth rates for juvenile Lemon Shark (Negaprion brevirostris) were used (~10–25 cm TL per year; Freitas et al. 2006; Tavares et al. 2020), estimating a YOY size of <70–85 cm TL. Given similar sizes-at-birth, maturity, and maximum size of both species, YOY Sharptooth Lemon Shark were here estimated as those <80 cm TL.
Between May 2006 and April 2007, visual surveys were conducted through an aboveground viewing platform, to determine the movements and estimate the abundance of Giant Guitarfish during their intertidal migration in Giralia Bay (southeastern Exmouth Gulf) (Penrose 2011). Up to 2,049 individuals were observed migrating between mangrove and cyanobacterial mat habitats during a single tidal cycle per observation site, with an estimated density of 42.3–42.4 individuals per m2 per hour. This equates to an estimated 4,382 Giant Guitarfish in Giralia Bay. Body sizes of all the visually sighted Giant Guitarfish in this study were not reported, but >69% of individuals which underwent sampling for stomach content analysis (n = 57) in the same study site were neonates and YOY (<40–45 cm TL). It is likely that the majority of visually sighted individuals were of similar body size based on similar habitat and shallow depths (<0.5 m depth) in the visually surveyed habitat. Large numbers of neonate and juvenile Giant Guitarfish have been reported in Urala Creek North and South with body sizes visually estimated to range between ~40–50 cm TL (Morgan et al. 2020). Using gillnets, individuals captured ranged between 46.2–79.9 cm TL (Morgan et al. 2020). The size-at-birth of this species is 38–40 cm TL (Kyne et al. 2019), and YOY are here assumed to be those <50 cm TL based on growth rates (White et al. 2014). Between 2007–2008, visual surveys were conducted in Ningaloo Lagoon in which aggregations of Giant Guitarfish were identified in nearshore areas (in a few cm of water depth) (Stevens et al. 2009). Aggregations of up to 50 individuals were recorded at Winderabandi Point, Mangrove Bay, Point Cloates, and Pelican Point. Two of these aggregations were comprised of neonates, and the aggregation at Winderabandi Point was comprised of individuals 40–100 cm TL. In total, 170 Giant Guitarfish were identified through visual surveys within the Ningaloo Lagoon (Stevens et al. 2009).
Giant Guitarfish records were collated from citizen science records and research surveys in this area. Unpublished citizen science records of large-bodied rhino rays were collated from databases held within the following organizations: Fin Focus Research, Sharks and Rays Australia, Sawfish Conservation Society, and Murdoch University’s Centre for Sustainable Aquatic Ecosystems. These records included newspaper articles relating to occasional captures, sightings/captures from snorkellers, divers, recreational fishers, and other recreational ocean users. Records generally included the date, location (exact or approximate), a size estimation, and occasionally sex (Bateman et al. 2024). Most submissions were recent (within the last 15 years); historical submissions (n = 9) were also included from prior to 2008. Targeted research surveys used gillnets with a 150 mm mesh size, cast nets, and capture by hand (e.g., Morgan et al. 2015, 2017). These surveys were conducted across multiple years (2011–2022) and seasons (Bateman et al. 2024). A total of 771 Giant Guitarfish were recorded (473 from scientific surveys, 298 from citizen science submissions). Of these records, the majority (n = 641) were of neonates, YOY, and older juveniles (<150 cm TL). Large aggregations of neonates, YOY, and juveniles have been reported across multiple years at several sites, including Winderabandi Point (Ningaloo Reef), Bay of Rest, and Giralia Bay (Exmouth Gulf) (Bateman et al. 2024). These aggregations are often dominated by YOY, numbering up to 80 individuals (RL Bateman pers. obs. 2025).
Between 2020–2021, gillnet surveys (150 mm mesh size, 60 m length) conducted in the southern Pilbara (Ashburton River mouth, Hooley Lagoon, Hooley Creek, and Four Mile Creek) have also recorded YOY (n = 12) and older juvenile (n = 4) Giant Guitarfish (Ingelbrecht et al. 2024a, 2024b; KO Lear & DL Morgan unpubl. data 2025). In April 2024, October 2024, and April 2025, visual and fishery-independent gillnet surveys (150 mm mesh size, 60 m length) were conducted within the southern and eastern Exmouth Gulf, ranging from Bay of Rest to Urala Creek South in this area (KO Lear et al. unpubl. data 2024–2025). During these surveys, 149 Giant Guitarfish were visually sighted from small vessels traversing the shallows in mangrove creeks and mudflat habitats, including aggregations of up to 20 individuals. Of these visually sighted individuals, at least 70 were likely to be YOY based on estimated sizes. An additional 21 individuals were physically captured in gillnets, of which 7 were <50 cm TL. Between April 2024–April 2025, aerial drone surveys have also been conducted throughout Exmouth Gulf in this area (S Gudge & K Kliska unpubl. data 2024–2025). A total of 1,251 Giant Guitarfish were sighted during these surveys, including aggregations of up to 35 individuals. Visual size estimates spanned neonates to adults; however, most individuals, particularly in aggregations, were YOY or small juveniles (K Kliska pers. obs. 2025).
Australian Whiprays have been commonly visually observed within mangrove areas during gillnet sampling in Exmouth Gulf in this area. This includes 127 individuals visually observed during gillnet surveys in the southern and eastern Exmouth Gulf in 2021, 2022, 2024, and 2025 (RL Bateman-John unpubl. data 2025; KO Lear et al. unpubl. data 2025). Small Australian Whiprays which are likely to be YOY based on visual estimates, are particularly common in the Bay of Rest, where they enter mangrove areas at high tide, and can be observed emigrating from the mangroves in large numbers (~3–10 individuals per ~5 min) as the tide drops (KO Lear pers. obs. 2025). Of 127 survey observations, 90 individuals were visually sized; 66 of these (73%) were <40 cm disc width (DW), consistent with YOY or a mix of YOY and older juveniles. Between September 2021 and May 2022, groups of 4–15 Australian Whiprays of various body sizes were visually observed during vessel-based surveys on nine occasions in the southwestern Exmouth Gulf (RL Bateman-John unpubl. data 2022). In April and November 2024, groups of 4–5 small Australian Whiprays were recorded on two occasions during gillnet surveys in the southwestern and northeastern Exmouth Gulf (KO Lear et al. unpubl. data 2024). In 2024–2025, drone surveys were conducted throughout the southern and eastern Exmouth Gulf (K Kliska & S Gudge unpubl. data 2024–2025). Australian Whiprays, both YOY and larger individuals, are frequently observed in large mixed-species assemblages in this area. Australian Whiprays are not readily captured during gillnet surveys which have been the dominant catch method used during scientific surveys in Exmouth Gulf. As a result, captures of this species are limited. The size-at-birth of Australian Whiprays is ~29 cm DW, and size-at-maturity for males is estimated at 112 cm DW (Last et al. 2016; White et al. 2017). No growth information is available for this species.
Broad Cowtail Rays have been commonly observed during gillnet sampling across the southern and eastern Exmouth Gulf in this area. During vessel-based surveys, at least three large aggregations of 10–15 small individuals were seen in the shallows, and 46 individuals were recorded in southern and eastern Exmouth Gulf in 2024–2025. Among 34 individuals with visual size estimates, 15 (44%) were estimated to be <40 cm DW, indicating a mix of YOY and small juveniles (KO Lear et al. unpubl. data 2025). Drone surveys across the southwestern, southern, and eastern gulf in 2024–2025 counted 255 Broad Cowtail Rays (K Kliska & S Gudge unpubl. data 2024–2025). Drone footage confirms the common presence of small individuals, likely to be YOY or older juveniles. In 2021–2022, at least 96 individuals were visually observed from vessels during gillnet surveys in the Bay of Rest and adjacent sites, within this area. Of 55 individuals visually estimated, 45 were likely YOY or juveniles (RL Bateman-John unpubl. data 2022). Small individuals that are likely to be YOY are also common within mixed-species assemblages in this area, and these individuals can be observed exiting the mangrove areas in the Bay of Rest on falling tides in large numbers (~3–5 per 5-min period; KO Lear pers. obs. 2025). Only three Broad Cowtail Rays were captured in gillnet surveys during 2021–2025 with one individual measuring <30 cm DW and likely a YOY. Broad Cowtail Rays were among the most frequently observed elasmobranchs in the Bay of Rest but are seldom captured in gillnets, the primary scientific survey method used in Exmouth Gulf. Size-at-birth is ~18 cm DW (Last et al. 2016); growth data are unavailable, precluding a precise YOY size threshold.
In April and October 2024–2025, gillnet surveys were conducted in Urala Creek South in Ningaloo & Exmouth Coast ISRA in this area (KO Lear unpubl. data 2025). Ten YOY and 14 juvenile Green Sawfish were reported in Urala Creek South in 2020 and 2022–2025 (RL Bateman-John unpubl. data 2025; KO Lear unpubl. data 2025). No adult Green Sawfish have been captured in this area. A YOY Green Sawfish tagged in Urala Creek South in April 2024 was recaptured there in October, with acoustic data showing ~10 months of residency until a cyclone in February 2025, and three age 1+ juveniles tagged in April and October 2024 also showed high residency until the same event (KO Lear, S Gudge, & RL Bateman-John unpubl. data 2025). Neonate and YOY Green Sawfish measure <90 and <100 cm TL, respectively (Lear et al. 2023).
CRITERION C
SUB-CRITERION C2 – FEEDING AREAS
Nyinggulu & Exmouth is an important feeding area for five shark and two ray species.
Ningaloo Reef Edge ISRA within the Nyinggulu & Exmouth area is an important feeding assemblage for Grey Reef Shark, Spinner Shark, and Dusky Shark. These species form assemblages in the area by aggregating to feed on schooling fish. Reports on social media have recorded these feeding aggregations with an average of at least 7 individuals (2–15) together on at least 14 events between November–April from 2022–2025. A diver who regularly visits this area 3–4 times per week for seven months each year reports observing these aggregations five times annually between November–April (DT Browne pers. obs. 2025).
Coral Bay ISRA within the Nyinggulu & Exmouth area is important for Tiger Shark feeding. Biologging tags were used to understand the three-dimensional fine-scale movement ecology of Tiger Sharks in this area (Andrzejaczek et al. 2019). Tags were deployed on 21 adult Tiger Sharks (265–380 cm TL) in April–May 2017 for durations of 5–48 h. Size-at-maturity is 226–305 cm TL for males and 250–350 cm TL for females (Ebert et al. 2021). Tags recorded both physical parameters such as depth and temperature, and, through the use of accelerometers, gyroscopes, and compasses, in situ measurements of animal trajectory and locomotion. Animal-borne-video enabled the recording of interactions with prey, specifically sea turtles (e.g., Loggerhead Turtle Caretta caretta). Sharks displayed tortuous movements (twists and turns) associated with prey searching for 27% of their tracks, and interactions with prey elicited varied responses including highly tortuous paths and burst movements. The cameras recorded 18 interactions with sea turtles, during which Tiger Sharks exhibited tortuous, circling movements and short bursts of acceleration near the turtles. These encounters likely represent prey investigations or exploratory hunting attempts. Several investigations of prey were immediately preceded by burst, stalking, and/or turning behaviours. Sea turtles are important grazers that occur in significant numbers at Ningaloo Reef (Preen et al. 1997). Previous studies have revealed that Tiger Sharks continuously oscillate through the water column, presumably to search for benthic prey on descent, and silhouetted air-breathing prey on ascent (Heithaus et al. 2002a; Nakamura et al. 2011). Sandflats may be preferred habitats for foraging by Tiger Sharks because prey here have less room to perform evasive manoeuvres and to escape (Heithaus et al. 2002b), increasing the efficiency of hunting. This is corroborated by reports from local ecotourism operators at Coral Bay who frequently observe Tiger Sharks hunting sea turtles in this area (Andrzejaczek et al. 2019). Tiger Sharks are also commonly observed by passive citizen scientists feeding on sea turtles in this area. In June 2007, August 2008, and May–June 2010, eight Tiger Sharks (185–399 cm TL) were satellite-tagged off the northwest coast of Australia (Ferreira et al. 2015). The movements of one shark were restricted to a relatively small space within this area and its surroundings for six months, showing long-term residency. Another two sharks, transmitting for 7 and 14 days, respectively, stayed within or nearby this area. These sharks were also likely using this area as a feeding ground.
Ningaloo Reef Edge ISRA within the Nyinggulu & Exmouth area is important for Whale Shark feeding. Research on Whale Sharks within this area spans decades with diverse methods used to examine seasonal aggregations, demographics, and foraging ecology. Overall, this area has been identified among the 25 largest aggregation sites for Whale Sharks globally (Araujo et al. 2022). Early work used aerial surveys and spotter planes to document seasonal aggregations, while hydro-acoustic surveys and water sampling linked their presence to zooplankton availability. Acoustic telemetry, archival, and satellite tagging later revealed fine- and broad-scale movements, complemented by vessel surveys, and photo-identification to describe population structure, site fidelity, and feeding ecology.
Initial observations of Whale Sharks in 1982, 1983, and 1985 had documented a seasonal aggregation in this area (Taylor 1989, 1994). Later studies established this area as a critical feeding ground for Whale Sharks. Aerial surveys (1989–1992) showed seasonal aggregations along the reef front, where sharks swam mainly parallel to the reef in turbid waters (Taylor 1996). The maximum density in any sector of the reef at any one time was four sharks per km2. Their arrival coincided with the annual mass coral spawning after the March–April full moon, when zooplankton levels rise (Simpson 1991; Taylor 1996; Rosser & Gilmour 2008). Hydro-acoustic surveys confirmed Whale Shark associations with frontal zones at reef passages, where zooplankton concentrated (Wilson et al. 2002). Feeding behaviour was frequently observed, including suction and ram filter feeding, with prey such as coral spawn, tropical krill Pseudeuphausia latifrons, mysids, and jellyfish (Norman 1999; Taylor 2007). Dense schools of the tropical krill, the dominant krill species in faecal DNA samples, were particularly important prey (Taylor 1996; Wilson et al. 2003a, 2003b; Jarman & Wilson 2004). Chlorophyll-a concentrations were also identified as drivers of distribution, as sharks were often sighted in higher-productivity regions (Sleeman et al. 2007). In 2018, vessel-based acoustic surveys and oceanographic profiling linked Whale Shark space use to prey distribution, showing that sharks concentrated feeding activity along reef gutters and pinnacles where copepods and krill were most abundant (D’Antonio et al. 2024).
Research in the mid-1990s showed Whale Shark aggregations are dominated by juveniles (~600–800 cm TL; range 400–1,200 cm TL; ~85% male). Females were rarer and smaller (mean, 620 cm TL). Photo-identification (1992–2004) recorded 159 sharks (74% male; 300–1,000 cm TL) and a super-population of ~300–500. In 2018, juveniles remained male-biased (68% of 400; 300–900 cm TL) (Norman 1999; Meekan et al. 2006; Norman & Stevens 2007; D’Antonio et al. 2024).
Photo-identification between 1992–2004 shows strong philopatry: 29% of 159 Whale Sharks were resighted within the same year, and 33 across years, typically after 1–3 years, with two after 12 years. One shark returned over 19 years (1995–2016). These shows that they repeatedly feed here for decades.
Telemetry studies on Whale Sharks in the area since the 1990s have confirmed characteristic surface-feeding behaviours and close association with the reef (Gunn et al. 1999; Wilson et al. 2001; Gleiss et al. 2011, 2013). Tour-operator data from 2006–2010 also documented a sharp rise in Whale Shark interactions (350% increase; n = 3,254), showing predictable seasonal aggregations between March–July, with encounters concentrated near reef passages within 3–6 km of the reef crest (Anderson et al. 2014). Fine-scale studies in 2018 revealed that tagged sharks used both surface waters and deeper zones (40–60 m), linking their diving behaviour to mesozooplankton distribution and local oceanographic features (D’Antonio et al. 2024).
Ningaloo & Exmouth Coast ISRA within the Nyinggulu & Exmouth area is important for Giant Guitarfish feeding. Between May 2006 and April 2007 (April–May and October–November), gill and fyke nets were used to capture Giant Guitarfish within mangrove and cyanobacterial mat habitats at three sites in Giralia Bay (Penrose 2011). Stomach contents and muscle samples (liver and muscle tissues) of 57 individuals with a body size of 39–108 cm TL, which corresponds to neonates, YOY, and older juveniles, were used for stomach and stable isotope analyses to estimate trophic dependency to the intertidal estuary habitat and ontogenetic dietary shifts of Giant Guitarfish (Penrose 2011).
Penaeid prawns (Western School Prawn Metapenaeus dalli; 72% Index of Relative importance; IRI) and brachyuran crabs (20% IRI) were the most important dietary categories for juvenile Giant Guitarfish. Penaeid prawns were present in 82.5% of all stomachs and made the greatest contribution to the total numbers and volumes of all dietary categories. This indicates that early life-stages of this ray has a narrow diet composed mainly of crustaceans. The stomach content data suggest dietary overlap between the two size classes (<60 cm [mostly neonates and YOY] and 60–100 cm TL), whereas the stable isotope data indicate that the primary production sources supporting Giant Guitarfish food webs changes with size (age). Size-at-birth is 38–40 cm TL (Kyne et al. 2019), and individuals <50 cm TL are classified as YOY based on early growth rates (White et al. 2014). Isotopic values of muscle tissue and liver tissue were consistent with a dependency on high intertidal cyanobacterial mat (30–76%) and seagrass-based (9–55%) food webs. Reliance on the high intertidal cyanobacterial mat was not reflected in either liver or muscle tissue until sufficient growth had occurred. At approximately 90 cm TL, the isotopic values (δ13C) of Giant Guitarfish tissues reach equilibrium with their post-natal benthic nearshore food resources. The carbon isotope composition of smaller individuals (<90 cm TL) reflects the maternal dietary influence from an offshore food web.
The mean number of Giant Guitarfish that undertook tidal migrations onto the high intertidal cyanobacterial mat varied between sites but ranged from 1,356 ± 271 to 1,060 ± 177 per hr in Ningaloo & Exmouth Coast ISRA within the Nyinggulu & Exmouth area (Penrose 2011). At one site, the maximum number of observed rays migrating between the habitats during the first hour of flood tide was 2,049 individuals. The mean density of Giant Guitarfish per m2 per hr on the cyanobacterial mat was similar between sites, ranging between 42.3 ± 4.4 and 42.4 ± 3.6 individuals. These visual surveys indicate that juvenile Giant Guitarfish undertake tidal migrations to the high intertidal cyanobacterial mats in high numbers to feed on a highly productive intertidal environment (Seckbach & Oren 2010; Penrose 2011).
No recent studies have examined the diet of Giant Guitarfish in this area; however, unpublished drone surveys continue to record large aggregations of YOY near or on cyanobacterial mats in the southern and eastern Exmouth Gulf (KO Lear et al. unpubl. data 2025). The shallow mudflats, mangroves, seagrass, and algal beds of eastern and southern Exmouth Gulf form key nursery habitats for juvenile prawns (Loneragan et al. 2013; Kangas et al. 2015). These productive areas support high abundances of Western King Prawn (Melicertus latisulcatus) and Tiger Prawn (Penaeus monodon), with central gulf waters yielding the greatest catches, surpassing other Pilbara trawl fisheries. This productivity, linked to extensive nursery grounds, likely sustains additional crustacean species dependent on similar habitats (Loneragan et al. 2013; Kangas et al. 2015; WAMSI 2025).
Ningaloo Reef Edge ISRA within the Nyinggulu & Exmouth area is important for Reef Manta Ray feeding. Reef Manta Rays have regularly and predictably used this area for feeding, attracting tourists since the early 1990s. Their reliable presence has led to a steady growth in manta tour operations, which can now accommodate over 130 passengers per day to swim with these rays (Venables et al. 2016). Ventral photographs of Reef Manta Rays sighted in this area have been catalogued since 2006 in a photo-identification image database along with metadata such as date and location of sighting (McGregor et al. 2019; Armstrong et al. 2020a). Photos were collected primarily by tourism operators who run in-water megafauna interaction tours year-round (2002–2018). As of 2015, long-term photo-identification studies have identified 800 individuals (Venables et al. 2016; Armstrong et al. 2020a; F McGregor unpubl. data 2015). The behaviour displayed by each individual when sighted was categorised (Germanov et al. 2019) and was informed by ancillary observations reported by tourism operators or trained observers, or through indications of behaviour evident in photographs. A total of 4,534 behavioural observations of Reef Manta Rays were recorded in this area (Armstrong et al. 2020a). Feeding was the most common behaviour with 56–80% of observations. Between March–May, large feeding aggregations of up to 70 individuals were recorded in Coral Bay (McGregor et al. 2019). The core resident population in Coral Bay consists of 40–50 individuals, mostly mature females (McGregor et al. 2019).
CRITERION C
SUB-CRITERION C5 – UNDEFINED AGGREGATIONS
Nyinggulu & Exmouth is important for undefined aggregations of one shark and four ray species.
Coral Bay ISRA within the Nyinggulu & Exmouth area is important for undefined aggregations of Blacktip Reef Shark. Between November and December of 2007–2009 and in August 2009, a total of 58 sharks were caught from the beach at Skeleton Bay within the area using hand reels and baited barbless hooks. For each individual, species, sex, and size (TL) were recorded and acoustic tags implanted for telemetry monitoring. An array of five acoustic receivers was deployed to monitor movement patterns of 36 Blacktip Reef Sharks in Skeleton Bay (Speed et al. 2011). Visual census surveys were also undertaken from the top of a sand dune located at the back of the beach (Speed et al. 2011). Observations of sharks in Skeleton Bay were noted and individuals counted hourly, when possible, between 08:00 and 17:00 h over 25 days in November/December 2008, 19 days in November/December 2009, four days in August 2009, and four days in February 2010. An ‘aggregation’ was defined as five or more individuals present in the same zone at the same time (as per Heupel & Simpfendorfer 2005).
Blacktip Reef Shark was the most abundant species caught in Skeleton Bay (n = 36; 62%) and forming the core of the multi-species reef shark assemblage (Speed et al. 2011). Telemetry detections reported that multiple tagged Blacktip Reef Sharks (adult males and females) were recorded on the same day and at the same time (~13:00–14:00 h). Visual census reported that assemblages were dominated by adult Blacktip Reef Shark, with up to 44 individuals counted in a single aggregation and a maximum of 74 across Skeleton Bay during November 2008. This species was present regularly throughout the year and observed in ~62% of 158 censuses. Individuals ranged between 88–144 cm TL. The average (± standard deviation) body length of tagged individuals ranged from 103 ± 0.16 cm TL to 129 ± 0.13 cm TL across years (2007–2009), indicating that most were mature adults. Size-at-birth is 45–64 cm TL (Ebert et al. 2021), male size-at-maturity is 130–145 cm TL and female size-at-maturity is 120–142 cm TL (Ebert et al. 2021). The sex ratio was skewed toward females (1.4:1.0), and numerous females displayed fresh mating scars and signs of pregnancy (distended abdomen), providing strong evidence of reproductive activity within the aggregation site. Courtship behaviour was also documented, and both adult males and gravid females were commonly seen together, suggesting that Skeleton Bay functions as a mating area. Juvenile Blacktip Reef Shark were consistently detected over multiple years, although neonates were not reported. Still, more information is needed to confirm that this area functions as a reproductive area. Aggregations of reef sharks in this area could be facilitating thermoregulation as they actively seek warmer waters to elevate body temperature (Speed et al. 2011, 2012).
Coral Bay is a recognised shark aggregation site. Skeleton Bay belongs to the shark dreaming of the Nyinggulu Ganyarjarrri people, the Traditional Owners of the Ningaloo Coast, who call it Nhuga Malinmayi, meaning the shark birthing place. It is recognised as a culturally sacred and natural nursery, where juvenile sharks use the sheltered coastal waters for protection until they mature and move offshore. This reflects the importance of this area for centuries. This knowledge is also supported by passive citizen-science posts from 2022–2024 and by the Western Australia Department of Biodiversity, Conservation and Attractions. More than 200 individuals—mostly Blacktip Reef Sharks—have been recorded in Skeleton Bay between October–March (CBET 2025; DBCA 2025; Tripadvisor 2025).
Ningaloo & Exmouth Coast ISRA within the Nyinggulu & Exmouth area is important for undefined aggregations of Australian Whipray, Broad Cowtail Ray, and Bluespotted Lagoon Ray. Various sources have recorded large aggregations of rays within the shallow mangrove-associated habitats in this area. This includes drone surveys conducted across the southern and eastern Exmouth Gulf between 2024–2025 (S Gudge & K Kliska unpubl. data 2024–2025); visual observations during fishery-independent gillnet surveys conducted in the southwestern Exmouth Gulf in 2021–2022 (RL Bateman-John unpubl. data 2022) and across the southern and eastern Exmouth Gulf in 2024–2025 (KO Lear et al. unpubl. data 2025); and various citizen science contributions. These have included single-species aggregations of 10–35 individuals of Broad Cowtail Rays, and Blue-spotted Fantail Rays, as well as large mixed-species assemblages generally including Australian Whiprays and Broad Cowtail Rays.
Between September 2021–May 2022, during fishery-independent gillnet surveys, groups of 4–15 Australian Whiprays of various body sizes were visually observed during vessel-based surveys on nine occasions in the southwestern Exmouth Gulf in Ningaloo & Exmouth Coast ISRA within the Nyinggulu & Exmouth area (RL Bateman-John unpubl. data 2022). In April and November 2024, groups of 4–5 Australian Whiprays were recorded on two occasions during gillnet surveys in the southwestern and northeastern Exmouth Gulf (KO Lear et al. unpubl. data 2024). In 2024–2025, drone surveys were conducted throughout the southern and eastern Exmouth Gulf (S Gudge & K Kliska unpubl. data 2024–2025). Australian Whiprays, both YOY and larger individuals, were frequently observed in large mixed-species assemblages in this area.
Between September 2021 and May 2022, during fishery-independent gillnet surveys, groups of 4–15 Broad Cowtail Rays were visually recorded on nine occasions in and near the Bay of Rest, and between April 2024 to April 2025, groups of 5–7 were noted resting or feeding on four occasions in Ningaloo & Exmouth Coast ISRA within the Nyinggulu & Exmouth area (KO Lear et al. unpubl. data 2024). Drone surveys between August 2024 and March 2025 documented groups of 4–11 resting and/or feeding on six occasions (S Gudge & K Kliska unpubl. data 2025). During vessel-based surveys, at least three large aggregations of 10–15 small individuals were seen in the shallows.
In January 2022, six subadult–adult Bluespotted Lagoon Rays were recorded in an aggregation near Wapet Creek (RL Bateman-John unpubl. data 2022). In November 2024, 10–15 juvenile and adult individuals were observed near Doole Island (KO Lear et al. unpubl. data 2024). Drone surveys in 2024–2025 recorded aggregations: 10 and 15 individuals, each within three minutes in August 2024, 10 individuals within six minutes in November 2024, and 47 individuals within ten minutes in April 2025 (S Gudge & K Kliska unpubl. data 2025). Across the 2024–2025 surveys, 85% of all records for this species were from these aggregations, with single individuals rarely observed.
For the mixed-species assemblages, in January 2022, during fishery-independent gillnet surveys, groups of 10–20 rays, consisting of Australian Whiprays and Broad Cowtail Rays were observed on three occasions in the southwestern Exmouth Gulf (RL Bateman-John unpubl. data 2022). Between April and October 2024, during drone surveys, groups of 4–13 unidentified whiprays, likely Australian Whiprays were recorded on three occasions (S Gudge & K Kliska unpubl. data 2024). In September 2021, April 2024, and March 2025, mixed-species assemblages of approximately 15–30 juveniles, including Australian Whiprays and Broad Cowtail Rays, were observed on several occasions in the northwestern Bay of Rest (KO Lear pers. obs. 2025). These aggregations are likely driven by either foraging (notably when adults are prevalent) or shallow-water refuge, with neonates, YOY, and juveniles of multiple species often mixing in extreme shallows at high tide.
Ningaloo Reef Edge ISRA within the Nyinggulu & Exmouth is important for undefined aggregations of Australian Cownose Rays. Aggregations have been regularly documented through passive citizen science within the area. Between 2017–2023, 12 social media reports recorded groups of at least 20 individuals each, with most aggregations composed of ~30 individuals (~20–100). In one of these reported observations, a potential courtship behaviour was recorded.
Further information is required to determine the nature and function of these aggregations.
CRITERION D
SUB-CRITERION D1 – DISTINCTIVENESS
Nyinggulu & Exmouth is important for distinct attributes of one shark species.
Coral Bay ISRA within the Nyinggulu & Exmouth area is important for distinct attributes of Grey Reef Sharks. This species regularly visits Asho’s Gap which functions as a cleaning station where divers and snorkelers report sharks being cleaned by Bluestreak Cleaner Wrasse (Labroides dimidiatus) and Moon Wrasse (Thalassoma lunare). The most common behaviour for potential shark clients was to cruise over the cleaning station, but some clients would hover (Coward 2017). During cleaning interactions, individuals slowly swim over the coral cleaning station and will adopt a vertical tail-stand pose with their mouth open. Usually, one cleaner wrasse will approach to remove parasites and dead tissue from their gills, mouth, and body surfaces for an average of 7.9 seconds (Coward 2017; Huisman & van Keulen 2017). This site is well-known by passive citizen scientists and researchers (Speed et al. 2012; Huisman & van Keulen 2017; AMA 2025; CBA 2025; Exmouth Resort 2025; SSI 2025). Sharks are seen year-round, either individually or in groups of up to 20 individuals. Aggregations however peak in summer and autumn when individuals are observed being cleaned (Coward 2017). This area is especially important as it is the only known cleaning stations for Grey Reef Sharks within the Eastern Indian Ocean. Globally, cleaning behaviour of Grey Reef Sharks has been reported in only two ISRAs, both in the Maldives (Southern Laamu Atoll ISRA and Laamu Maavah Kandu ISRA; Jabado et al 2023).
CRITERION D
SUB-CRITERION D2 – DIVERSITY
Nyinggulu & Exmouth sustains a high diversity of Qualifying Species (39 species). This exceeds the regional diversity threshold (26 species) for the Australia and Southeast Indian Ocean.
The regular presence of Qualifying Species has been documented through records from fishery-independent surveys, Baited Remote Underwater Video System (BRUVS) surveys, visual observations from vessel-based and shore-based surveys, snorkel and SCUBA underwater visual surveys, aerial surveys (drones and spotter planes), citizen science, environmental DNA (eDNA) surveys, animal-borne-video, satellite and acoustic tracking, photo-identification, biologging, and traditional ecological knowledge (Taylor 1996, 2007; Gunn et al. 1999; Norman 1999; Wilson et al. 2001; Meekan et al. 2006; Stevens et al. 2009; Gleiss et al. 2011, 2013; Penrose 2011; Speed et al. 2011, 2012; O’Shea et al. 2013; Anderson et al. 2014; Cerutti-Pereyra et al. 2014; Chin 2014; Ferreira et al. 2015; Ashe 2016; Norman & Morgan 2016; Hoschke & Whisson 2016; Oh et al. 2016, 2017; Braccini et al. 2017; Coward 2017; Andrzejaczek et al. 2018, 2019; McGregor et al. 2019; Araujo et al. 2020, 2022, 2024; Armstrong et al. 2020a, 2020b; Morgan et al. 2020; Huveneers et al. 2021; Pillans et al. 2021; Lester et al. 2022; Sprogis & Parra 2022; Hoschke et al. 2023; Bateman et al. 2024; D’Antonio et al. 2024; Ingelbrecht et al. 2024a, 2024b; Lear et al. 2024; ALA 2025; GlobalArchive 2025; The Fish Collective 2025; Marine Futures Lab unpubl. data 2009–2021; FinPrint unpubl. data 2014–2015; DBCA unpubl. data 2019; RL Bateman-John unpubl. data 2021–2025; J Bonesso & J Pearce unpubl. data 2024; S Gudge & K Kliska unpubl. data 2024–2025; KO Lear et al. unpubl. data 2024–2025; M Braccini unpubl. data 2025; KO Lear & DL Morgan unpubl. data 2025; B D’Antonio pers. comm. 2023; DT Browne pers. obs. 2025). The combination of these methods showed the regular presence of these species in this area.
Fishery-independent surveys conducted between 2002–2025 using gillnets, longlines, hand nets, and spear guns recorded the presence of Nervous Shark, Silvertip Shark, Pigeye Shark, Copper Shark, Blacktip Shark, Blacktip Reef Shark, Sandbar Shark, Sand Tiger Shark, Tawny Nurse Shark, Sharptooth Lemon Shark, Milk Shark, Scalloped Hammerhead, Great Hammerhead, Indo-Pacific Leopard Shark, Whitetip Reef Shark, Spotted Eagle Ray, Ornate Eagle Ray, Giant Guitarfish, Australian Whipray, Broad Cowtail Ray, Pink Whipray, Jenkins’ Whipray, Green Sawfish, Bowmouth Guitarfish, Bottlenose Wedgefish, Blotched Fantail Ray, Bluespotted Lagoon Ray, Porcupine Ray, and Mangrove Whipray (Penrose 2011; O’Shea et al. 2013; Oh et al. 2017; Lear et al. 2024; Ingelbrecht et al. 2024a, 2024b; M Braccini unpubl. data 2025; RL Bateman-John unpubl. data 2025; KO Lear et al. unpubl. data 2024–2025).
Synthesised data from the Global Archive and The Fish Collective for BRUVS surveys were extracted after quality control was undertaken using CheckEM (GlobalArchive 2025; The Fish Collective 2025; Gibbons et al. in press). Also, BRUVS surveys from other sources were used. More than 1,000 BRUVS conducted in 2003–2024 in this area show the presence of the following species over multiple years: Silvertip Shark, Pigeye Shark, Copper Shark, Blacktip Shark, Sandbar Shark, Tawny Nurse Shark, Milk Shark, Scalloped Hammerhead, Great Hammerhead, Indo-Pacific Leopard Shark, Whitetip Reef Shark, Giant Guitarfish, Spinetail Devil Ray, Broad Cowtail Ray, Pink Whipray, Jenkins’ Whipray, Bowmouth Guitarfish, Australian Cownose Ray, Bottlenose Wedgefish, Blotched Fantail Ray, and Mangrove Whipray (Oh et al. 2016; Lester et al. 2022; FinPrint unpubl. data 2014–2015; DBCA unpubl. data 2019; Marine Futures Lab unpubl. data 2009–2021).
Visual observations from vessel-based and shore-based surveys were used to record the regular presence of Blacktip Reef Shark, Whitetip Reef Shark, Whale Shark and Spotted Eagle Ray (Norman 1999; Taylor 2007; Speed et al. 2011; Anderson et al. 2014; KO Lear et al. unpubl. data 2024–2025).
Snorkel and SCUBA underwater visual surveys conducted between 2007–2008 have recorded the regular presence of Silvertip Shark, Grey Reef Shark, Pigeye Shark, Copper Shark, Blacktip Shark, Blacktip Reef Shark, Sandbar Shark, Sand Tiger Shark, Sand Tawny Nurse Shark, Scalloped Hammerhead, Great Hammerhead, Indo-Pacific Leopard Shark, Whitetip Reef Shark, Spotted Eagle Ray, Giant Guitarfish, Broad Cowtail Ray, Pink Whipray, Jenkins’ Whipray, Bottlenose Wedgefish, Blotched Fantail Ray, and Mangrove Whipray (Stevens et al. 2009; Speed et al. 2011; Ashe 2016; Coward 2017; Hoschke & Whisson 2016; Hoschke et al. 2023).
Aerial surveys between 2024–2025 were used to record the presence Indo-Pacific Leopard Shark, Whale Shark, Spotted Eagle Ray, Bottlenose Wedgefish, Giant Guitarfish, Australian Whipray, Broad Cowtail Ray, Pink Whipray, Bluespotted Lagoon Ray and Porcupine Ray (Taylor 1996; Morgan et al. 2020; Sprogis & Parra 2022; KO Lear et al. unpubl. data 2024–2025; S Gudge & K Kliska unpubl. data 2024–2025).
Citizen science has also recorded the regular presence of Silvertip Shark, Grey Reef Shark, Pigeye Shark, Copper Shark, Spinner Shark, Blacktip Shark, Blacktip Reef Shark, Dusky Shark, Sandbar Shark, Sand Tiger Shark, Tawny Nurse Shark, Milk Shark, Scalloped Hammerhead, Great Hammerhead, Indo-Pacific Leopard Shark, Whitetip Reef Shark, Whale Shark, Spotted Eagle Ray, Ornate Eagle Ray, Giant Guitarfish, Reef Manta Ray, Oceanic Manta Ray, Spinetail Devil Ray, Shortfin Devil Ray, Broad Cowtail Ray, Pink Whipray, Jenkins’ Whipray, Green Sawfish, Bowmouth Guitarfish, Australian Cownose Ray, Bottlenose Wedgefish, Blotched Fantail Ray, Porcupine Ray, and Mangrove Whipray (Chin 2014; Araujo et al. 2020, 2024; ALA 2025; Bateman et al. 2024; DT Browne pers. obs. 2025).
eDNA surveys in 2014 were used to record the presence of Tawny Nurse Shark, Milk Shark, Great Hammerhead, Indo-Pacific Leopard Shark, Whitetip Reef Shark, Spotted Eagle Ray, and Porcupine Ray (J Bonesso & J Pearce unpubl. data 2024).
Acoustic telemetry was used between 2007–2020 and 2022 to record the movements of Blacktip Reef Shark, Copper Shark, Sandbar Shark, Sharptooth Lemon Shark, Broad Cowtail Ray, and Bottlenose Wedgefish (Cerutti-Pereyra et al. 2014; Speed et al. 2011; Braccini et al. 2017; Huveneers et al. 2021; Pillans et al. 2021; Lear et al. 2024). Sandbar Sharks have been detected for up to four years showing long-term residency or repeated returns to the area (Braccini et al. 2017).
Satellite telemetry has been used to study the habitat use and regular presence of the Tiger Shark and Whale Shark (Gunn et al. 1999; Wilson et al. 2001; Gleiss et al. 2011, 2013; Ferreira et al. 2015; D’Antonio et al. 2024).
Photo-identification has been used to study the regular presence of the Whale Shark, Reef Manta Ray and Oceanic Manta Ray (Meekan et al. 2006; Norman & Morgan 2016; McGregor et al. 2019; Armstrong et al. 2020a, 2020b; Araujo et al. 2022).
Biologging tags were used to understand the movement ecology of the Sandbar Shark and Tiger Shark in this area (Andrzejaczek et al. 2018, 2019).
Traditional ecological knowledge from the Nyinggulu Ganyarjarrri people has provided evidence of the historical use of this area by the Blacktip Reef Shark.
Download factsheet
SUBMIT A REQUEST
ISRA SPATIAL LAYER REQUEST
To make a request to download the ISRA Layer in either a GIS compatible Shapefile (.shp) or Google Earth compatible Keyhole Markup Language Zipped file (.kmz) please complete the following form. We will review your request and send the download details to you. We will endeavor to send you the requested files as soon as we can. However, please note that this is not an automated process, and before requests are responded to, they undergo internal review and authorization. As such, requests normally take 5–10 working days to process.
Should you have questions about the data or process, please do not hesitate to contact us.